Therapeutic Areas
With over 30 years of experience and a proven track record in developing new formulations and registering them worldwide, BTG is a leading manufacturer of biopharmaceutical products.
Our company’s in-house products are marketed worldwide by various Marketing and Sales subsidiaries of the Ferring Pharmaceuticals Group. Our product line has gained regulatory approval in the United States, all member states of the European Union, Israel and in a growing number of other countries.
Reproductive Health
Up to a quarter of all couples may experience problems in conceiving a child, a figure that appears to be rising partly due to the trend to delay pregnancy until an older age. The World Health Organization estimates that there are about 60 to 80 million cases of infertility around the world.
Treatment of infertility is one of Ferring’s key areas of expertise and we are firmly committed to progress in the field of Assisted Reproductive Technology (ART).
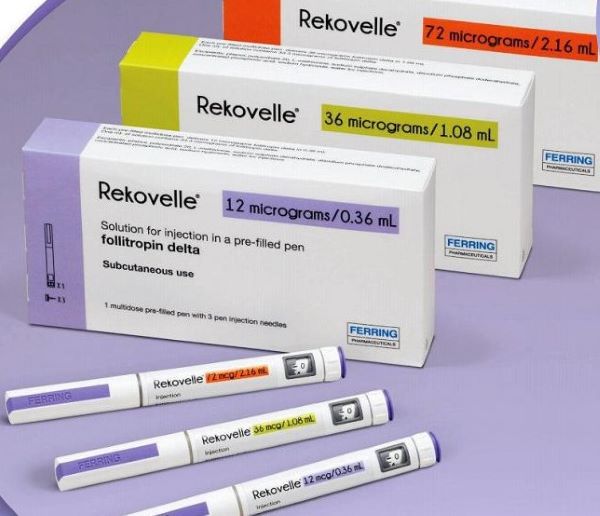
REKOVELLE® (follitropin delta), a human recombinant follicle stimulating hormone (human rFSH), for use in controlled ovarian stimulation for the development of multiple follicles in women undergoing fertility treatment.
Rekovelle is available as a solution for injection, contained in a cartridge to be used with Rekovelle injection pen.
REKOVELLE ® has regulatory approval in more than 70 countries, and is marketed in Europe and Asia
Endocrinology
Many children are smaller than average during childhood yet end up reaching a normal adult height. But for some, a more normal adult height will not be achieved without treatment with supplemental human growth hormone (hGH). These include children with Growth Hormone Deficiency (GHD) and Turner’s syndrome.
One out of 2,500 to 5,000 children is born with a deficient growth hormone production resulting in impaired physical development, and another one in 2,000 to 2,500 girls are born with the genetic defect Turner’s syndrome. Both disorders may result in decreased final height, which may be improved by supplemental somatropin.
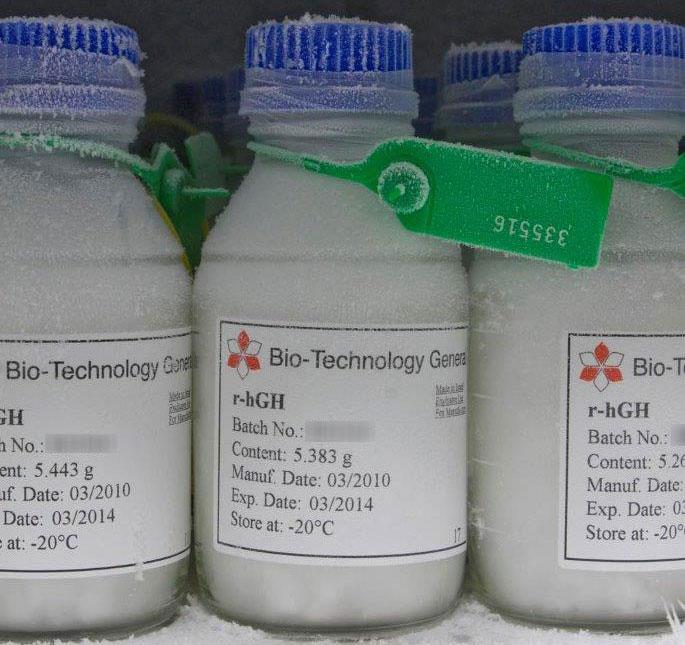
Recombinant Human Growth Hormone (Somatropin)
BTG’s recombinant human growth hormone was developed in the 1980s, first gained regulatory approval in 1988. It is identical to natural human growth hormone synthesized by the pituitary gland.

Orthopaedics
Ferring’s product EUFLEXXA® (1% sodium hyaluronate) is an ultra-high purity hyaluronate, called hyaluronic acid (HA). HA is the natural substance found in the fluid that surrounds a healthy knee joint and helps cushion, lubricate, and protect the knee as it moves throughout the day.
BTG’s NaHA is a highly purified, high molecular weight preparation, and, in contrast to other NaHA products extracted from avian sources, is produced using an efficient and proprietary bacterial fermentation process.
EUFLEXXA® has regulatory approval in more than 30 countries, and is marketed in North America, Europe and Asia. In Israel, the product is marketed under the trade name Arthrease® by Ferring Pharmaceuticals Ltd.
EUFLEXXA® is injected into the knees of osteoarthritis sufferers to improve movement and reduce pain.
More about Ferring's Orthopaedics Products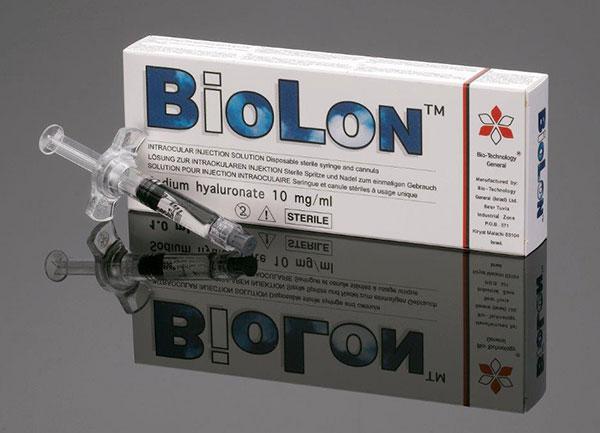
Ophthalmology
Biolon®, first introduced in 1993, is an injectable system containing 1% fermentation-derived sodium hyaluronate (NaHA) used as a viscoelastic aid in cataract and other ophthalmic surgery.
NaHA is a naturally occurring polysaccharide, ubiquitous in nature and chemically identical in all species. It is found in the vitreous and aqueous humor of the eye, in the synovial fluid surrounding knee and other joints, in skin and in other organs.
This high molecular weight, ultra-pure sodium hyaluronate, is manufactured in Bio-Technology General’s Be’er Tuvia facility using an efficient biosynthetic process. It is used to lubricate the eye and maintain low intraocular pressure while facilitating surgery to reduce trauma to the corneal endothelium.
Please note that products information presented on this website is intended only as a brief summary for the visitor’s convenience in relation to the activities of the Ferring Group. Not all products or indications are licensed in every country and may be subject to further local variations. For specific product information please consult the summary of product characteristics (SPC). For advice on medical issues please consult your local medical practitioner.
